6 results
Effects of a hard stop for C. difficile testing: Provider uptake and patient outcomes
- Danielle Doughman, David Weber, Nikolaos Mavrogiorgos, Shelley Summerlin-Long, Michael Swartwood, Alexander Commanday, Lisa Stancill, Nicholas Kane, Emily Sickbert-Bennett Vavalle
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) is a serious healthcare-associated infection responsible for >12,000 US deaths annually. Overtesting can lead to antibiotic overuse and potential patient harm when patients are colonized with C. difficile, but not infected, yet treated. National guidelines recommend when testing is appropriate; occasionally, guideline-noncompliant testing (GNCT) may be warranted. A multidisciplinary group at UNC Medical Center (UNCMC) including the antimicrobial stewardship program (ASP) used a best-practice alert in 2020 to improve diagnostic stewardship, to no effect. Evidence supports use of hard stops for this purpose, though less is known about provider acceptance. Methods: Beginning in May 2022, UNCMC implemented a hard stop in its electronic medical record system (EMR) for C. difficile GNCT orders, with exceptions to be approved by an ASP attending physician. Requests were retrospectively reviewed May–November 2022 to monitor for adverse patient outcomes and provider hard-stop compliance. The team exported data from the EMR (Epic Systems) and generated descriptive statistics in Microsoft Excel. Results: There were 85 GNCT orders during the study period. Most tests (62%) were reviewed by the ASP, and 38% sought non-ASP or no approval. Of the tests reviewed by the ASP, 33 (62%) were approved and 20 (38%) were not. Among tests not approved by the ASP, no patients subsequently received CDI-directed antibiotics, and 1 patient (5%) warranted same-admission CDI testing (negative). Of tests that circumvented ASP review, 18 (56%) ordering providers received a follow-up email from an associate chief medical officer to determine the rationale. No single response type dominated: 3 (17%) were unaware of the ASP review requirement, 2 (11%) indicated their patient’s uncharted refusal of laxatives, 2 (11%) indicated another patient-specific reason. Provider avoidance of the ASP approval mechanism decreased 38%, from 53% of noncompliant tests in month 1 to 33% of tests in month 6. Total tests orders dropped 15.5% from 1,129 during the same period in 2021 to 954 during the study period (95% CI, 13.4%–17.7%). Compliance with the guideline component requiring at least a 48-hour laxative-free interval prior to CDI testing increased from 85% (95% CI, 83%–87%) to 95% (95% CI, 93%–96%). CDI incidence rates decreased from 0.52 per 1,000 patient days (95% CI, 0.41–0.65) to 0.41 (95% CI, 0.32–0.53), though the change was neither significant at P = .05 nor attributable to any 1 intervention. Conclusions: Over time and with feedback to providers circumventing the exception process, providers accepted and used the hard stop, improving diagnostic stewardship and avoiding unneeded treatment.
Disclosures: None
Examining CLABSI rates by central-line type
- Lauren DiBiase, Shelley Summerlin-Long, Lisa Stancill, Emily Sickbert-Bennett Vavalle, Lisa Teal, David Weber
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s48-s49
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Central-line–associated bloodstream infections (CLABSIs) are linked to increased morbidity and mortality, longer hospital stays, and significantly higher healthcare costs. Infection prevention guidelines recommend line placement in specific insertion locations over others because of the relative risk of infection. The purpose of this study was to assess CLABSI rates by line type to determine whether some central lines had a lower risk of infection and should be recommended over others given similar clinical indications. Methods: At UNC Hospitals, data were obtained on central lines across a 3-year period (FY20–FY22) from the EMR (Epic Systems). Central lines were categorized as apheresis catheters, CVC lines (single, double, or triple lumen), hemodialysis catheters, introducer lines, pulmonary artery (PA) catheters, PICC lines (single, double, or triple lumen), port-a-catheters, trialysis catheters, or umbilical lines. The line type(s) associated with each CLABSI during the same period were recorded, and CLABSI rates by line type per 1,000 central-line days were calculated using SAS software. If an infection had >1 central-line device type associated, the infection was counted twice when calculating the CLABSI rate by line type. We calculated 95% CIs for each point estimate to assess for statistically significant differences in rates by line type. Results: During FY20–FY22, there were 264,425 central-line days and 458 CLABSIs, for an overall CLABSI rate of 1.73 CLABSIs per 1,000 central-line days. Also, 16% of patients with a CLABSI had >1 type of central line in place. Stratified data on CLABSI rates by each central-line type is presented in the Figure. CLABSI rates were highest in patients with apheresis lines (6.22; 95% CI, 3.96–9.35) and PA catheters (6.22; 95% CI, 3.54–10.20), and the lowest CLABSI rates occurred in patients with PICC lines (1.44; 95% CI, 1.19–1.73) and port-a-catheters (1.14; 95% CI, 0.89, 1.45). For both CVC and PICC lines, as the number of lumens increased from single to triple, CLABSI rates increased, from 0.91 to 2.63 and from 0.57 to 1.20, respectively. Conclusions: At our hospital, different types of central lines were associated with statistically higher CLABSI rates. Additionally, a higher number of lumens (triple vs single) in CVC and PICC lines were also associated with statistically higher CLABSI rates. These findings reinforce the importance of considering central-line type and number of lumens to minimize risk of CLABSI while ensuring that patients have the best line type based on their clinical needs.
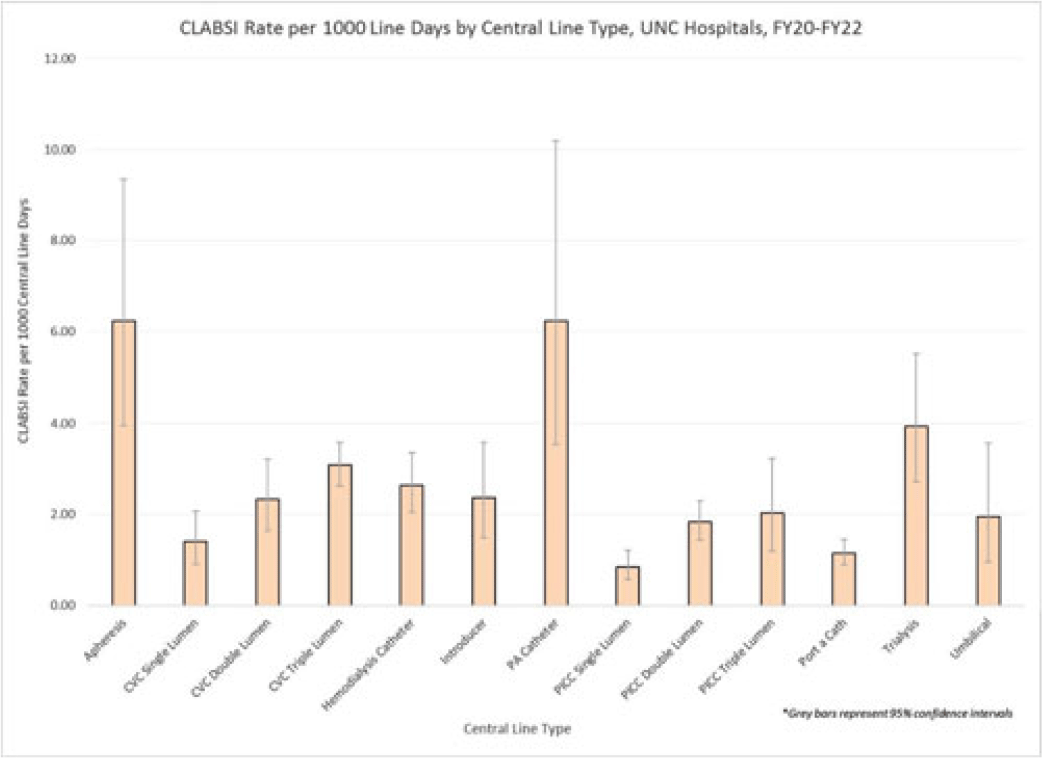
Disclosures: None
SARS-CoV-2 N95 contamination worn under a face shield, via medical mask surrogate, in healthcare providers treating COVID-19
- Amanda Graves, Bobby Warren, Aaron Barrett, Sarah Lewis, Becky Smith, David Weber, Emily Sickbert-Bennett Vavalle, Deverick Anderson
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s37-s38
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: SARS-CoV-2 N95 mask contamination in healthcare providers (HCPs) treating patients with COVID-19 is poorly understood. Method: We performed a prospective observational study of HCP N95 respirator SARS-CoV-2 contamination during aerosol-generating procedures (AGPs) on SARS-CoV-2–positive patients housed in a COVID-19–specific unit at an academic medical center. Medical masks were used as surrogates for N95 respirators to avoid waste and were worn on top of HCP N95 respirators during study AGPs. Study masks were provided to HCPs while donning PPE and were retrieved during doffing. Additionally, during doffing, face shields were swabbed with Floq swabs premoistened with viral transport media (VTM) prior to disinfection. Medical masks were cut into 9 position-based pieces, placed in VTM, vortexed, and centrifuged (Fig. 1). RNA extraction and RT-PCR were completed on all samples. RT-PCR–positive samples underwent cell culture infection to detect cytopathic effects (CPE). Contamination was characterized by mask location and front and back of face shields. Patient COVID-19 symptoms were collected from routine clinical documentation. Study HCPs completed HCP-role–specific routine care (eg, assessing, administering medications, and maintaining oxygen supplementation) while in patient rooms and were observed by study team members. Results: We enrolled 31 HCPs between September and December 2021. HCP and patient characteristics are presented in Table 1. In total, 330 individual samples were obtained from 31 masks and 26 face shields among 12 patient rooms. Of the 330 samples, 0 samples were positive for SARS-CoV-2 via RT-PCR. Positive controls were successfully performed in the laboratory setting to confirm that the virus was recoverable using these methods. Notably, all samples were collected from HCPs caring for COVID-19 patients on high-flow, high-humidity Optiflow (AGP), with an average of 960 seconds (IQR, 525–1,680) spent in each room. In addition to Optiflow and routine care, study speech pathologists completed an additional AGP of fiberoptic endoscopic evaluation of swallowing. Notably, 29 (94%) of 31 study HCP had physical contact with their patient. Conclusions: Overall, mask contamination in HCPs treating patients with COVID-19 undergoing AGPs was not detectable while wearing face shields, despite patient contact and performing AGP.
Funding: None
Disclosures: None


assessment of Vaccine Interest in Unvaccinated COVID-19–positive inpatients
- Lisa Stancill, Lauren DiBiase, Emily Sickbert-Bennett Vavalle
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s42
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Although vaccine hesitancy has been an issue for many years, it has become a major point of contention in the effort to mitigate the COVID-19 pandemic. In August 2021, a large academic medical facility began capturing the vaccination status of admitted COVID-19–positive patients, as well as their interest in the COVID-19 vaccine. We performed a descriptive analysis on the characteristics of unvaccinated patients who contracted COVID-19 and their interest in receiving the COVID-19 vaccine. Methods: Patient history and physical (H&P) notes and demographic data were collected using the internal data warehouse sourced from the electronic medical record for all SARS-COV-2–positive inpatient admissions to UNC Medical Center and UNC Chatham from August 1, 2021, to January 11, 2022. Manual chart reviews of progress notes were completed for patients whose history was not recorded in the initial H&P. Demographic data were summarized by vaccine status overall and by interest in COVID-19 vaccine among unvaccinated patients. We performed χ2 to determine demographic differences between the interested and uninterested unvaccinated groups. Results: In total, 536 patients were admitted with COVID-19 from August 1, 2021, to January 11, 2022. Of these, 15% were fully vaccinated (2 doses mRNA plus 1 dose J&J); 5.4% were partially vaccinated; 75.7% were unvaccinated; and 2.9% had an unknown vaccination status. Demographic characteristics are presented in Table 1. The most common demographics were consistent among the fully vaccinated and unvaccinated groups, with the exception of sex and age group (Table 1). For those whose interest data were available (n = 164), 34% were uninterested in receiving the COVID-19 vaccine. Importantly, race and age were statistically significantly different (P < .05) between the unvaccinated interested and unvaccinated uninterested groups. Conclusions: Even after experiencing COVID-19 firsthand and being hospitalized, some people who remain uninterested in receiving the COVID-19 vaccine. This population had a statistically higher proportion of white and older individuals than the unvaccinated interested group. Recommendations from healthcare providers might not be effective in persuading this population to be vaccinated. Instead, grassroots alternatives might be more successful. Additional analysis should be considered on whether patients who expressed interest in COVID-19 vaccine received immunization.
Funding: None
Disclosures: None
Building on the Foundation of a Sustainable Hand Hygiene Program During the COVID-19 Pandemic
- Lisa Stancill, Emily Sickbert-Bennett Vavalle, Lauren DiBiase
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s63-s64
-
- Article
-
- You have access Access
- Open access
- Export citation
-
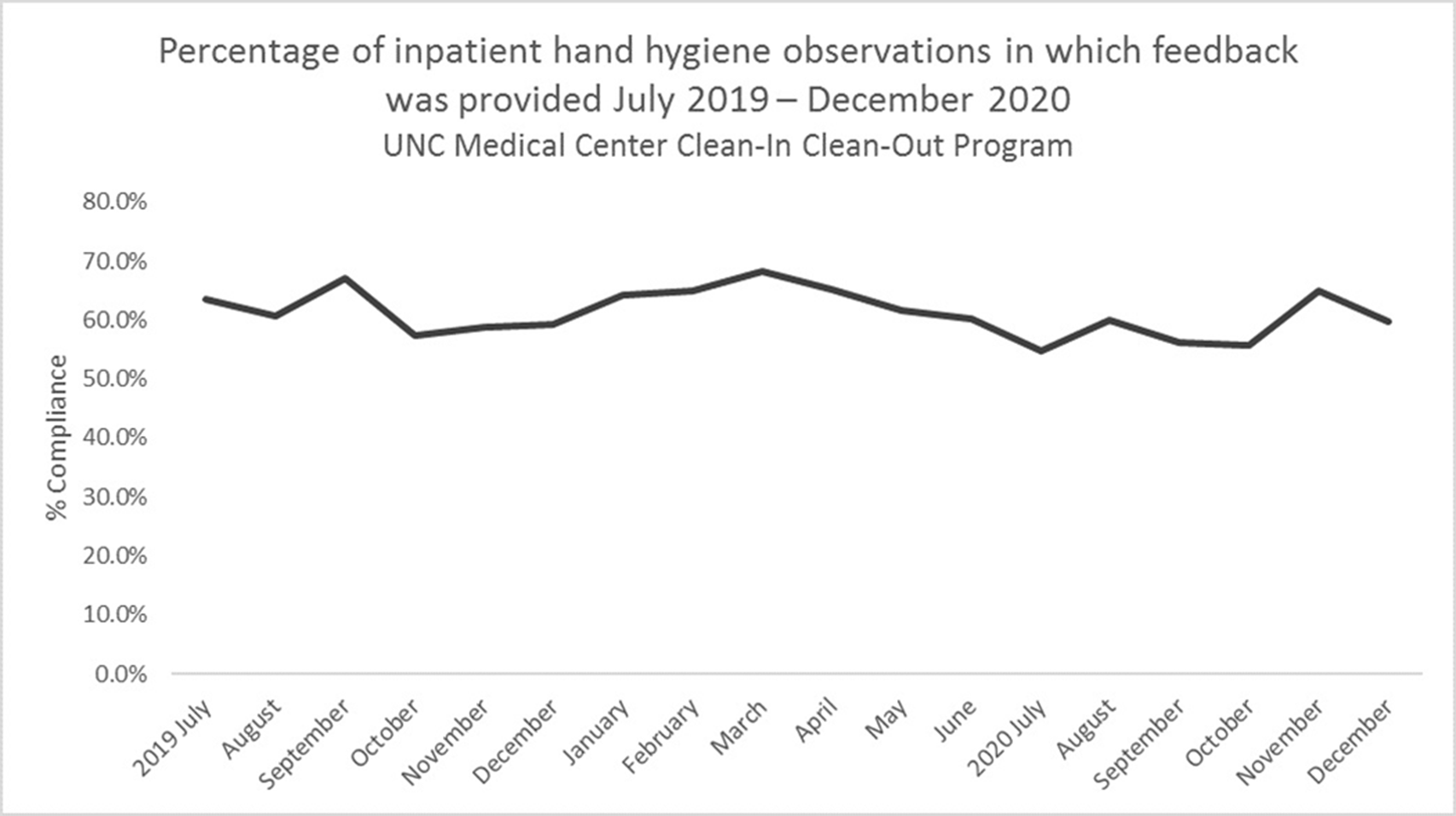
Background: Hand hygiene is essential to preventing the spread of disease in hospitals. Renewed emphasis has been placed on hand hygiene during the COVID-19 pandemic. We investigated whether UNC Medical Center’s well-established Clean-In Clean-Out (CICO) program for hand hygiene observations was sustainable throughout a public health and healthcare crisis and whether the COVID-19 pandemic had an effect on hand hygiene compliance. Methods: UNC Medical Center utilizes a crowd-sourced hand-hygiene audit application, CICO, to track hand-hygiene observations, compliance, and feedback. This application encourages participation from all staff and promotes providing real-time feedback in the form of a compliment or reminder when performing hand hygiene observations. During this evaluation, hand hygiene data were queried from the CICO application on the number of observations performed, hand hygiene compliance percentage, and feedback compliance percentage from July 2019 to December 2020. Hand hygiene data were compared to patient volumes in different care settings and the number of hospitalized patients being treated for COVID-19. Results: Initial increases in hand hygiene observations, compliance, and feedback were detected in the months leading up to UNC Medical Center receiving its first SARS-CoV-2–positive patient. Observations were highest when patient volumes were low due to closed clinics and restrictions on elective surgeries (Figure 1). When patient volumes returned to pre–COVID-19 levels coupled with treating more COVID-19 patients, the number of observations and compliance rate metrics declined. Feedback compliance percentage remained relatively stable through the entire period (Figure 2). Conclusions: Despite the additional strain on healthcare staff during COVID-19, the CICO model was a sustainable method to track hand hygiene observations and compliance. Notably, however, engagement was highest when patient census was lower, demonstrating that operating at a high capacity is not beneficial for patient safety. Due to the success and sustainment of the CICO program, UNC Medical Center used this model to create a Mask-On Mask-Up campaign to engage staff to submit observations, track compliance, and encourage feedback to promote the appropriate use of masks during COVID-19.
Funding: No
Disclosures: None
Figure 1.
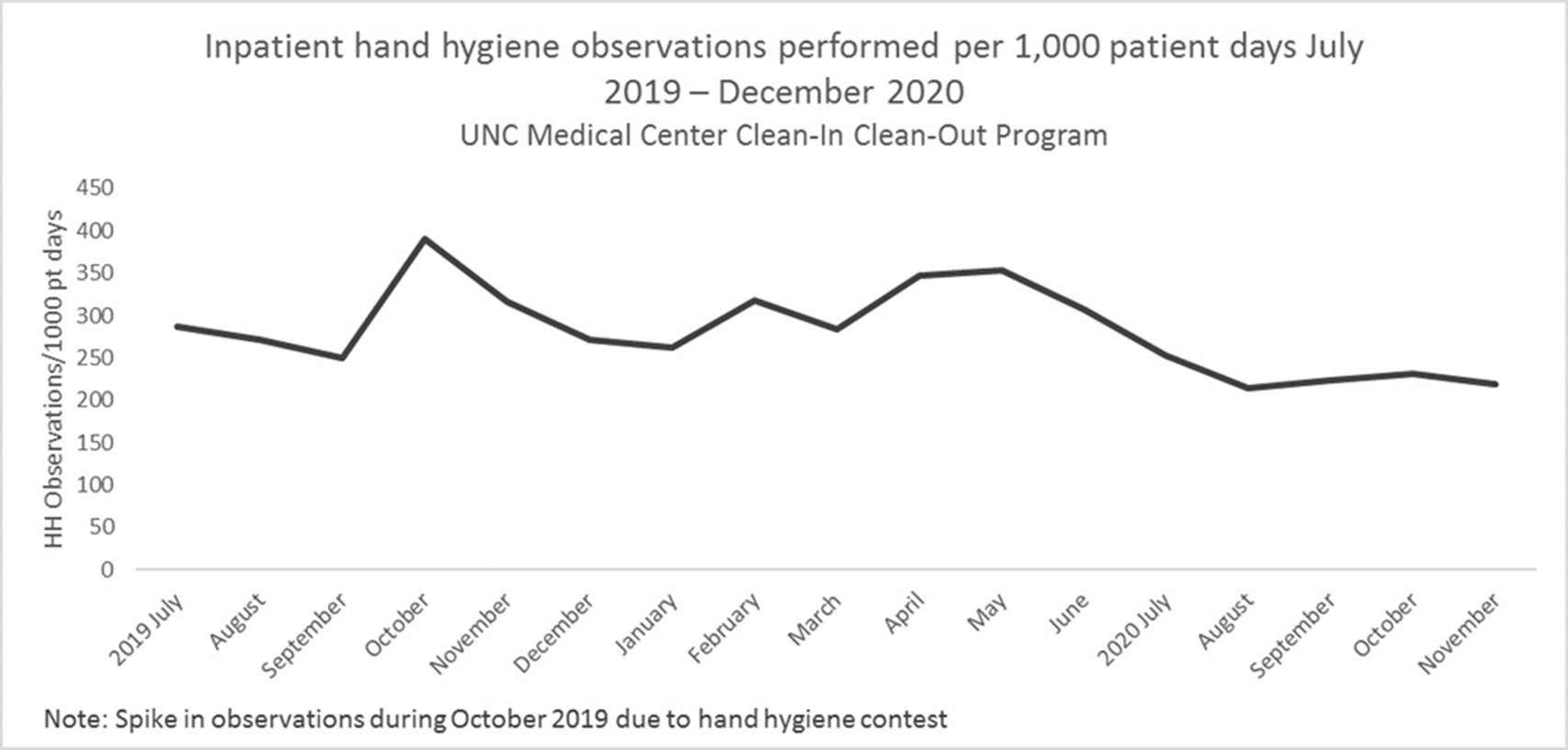
Figure 2.
Using a Quality-Driven Approach to Maintain an N-95 Respirator Supply During a Pandemic-Driven Global Shortage
- Amy Selimos, Mark Buchanan, Lauren DiBiase, Stephen Dean, Pat Boone, Nicholas Shaheen, Emily Sickbert-Bennett Vavalle, Beth Willis
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 1 / Issue S1 / July 2021
- Published online by Cambridge University Press:
- 29 July 2021, pp. s48-s49
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Reports of hospitals overwhelmed by COVID-19 patients created severe shortages of personal protective equipment (PPE). In this large academic medical system, we used a systematic team approach to proactively maintain an adequate PPE supply. The team consisted of staff from multiple departments including infection prevention, environmental health and safety, operational efficiency, and supply chain. The healthcare system solicited donations of PPE, and our team was tasked with developing a sustainable method to provide healthcare workers with safe and effective N-95 respirators. Respirators are normally fitted to our 6,000+ healthcare workers through a fit-testing process using 4 models of N-95s. We received >60 models, many in small quantities, posing a new level of complexity that prevented use of our typical fit-testing method. Methods: Donated respirators were manually verified on the CDC/NIOSH website to validate approval or approved alternative. A categorization system was developed, and respirators were sorted based on quality, style, and condition. User seal checks replaced qualitative fit testing due to the uncertain and quickly changing respirator supply. Staff were educated about the importance of performing a seal check to evaluate respirator fit and were provided instructions for what to do if they failed a seal check. We performed limited quantitative fit testing on a small group previously fit tested to 1 of the 4 models of N-95s normally stocked to identify the most effective alternative respirators to serve as substitute N-95s. Results: We were able to provide staff with new N-95s and delay the release of reprocessed N-95s. Overall, 18 models of respirators were tested on staff for filtration effectiveness and fit. We deemed 61% masks to be of last resort, and these were not released. We determined that 39% were acceptable as an alternative for at least 1 of our usual respirator models. However, only 3 models (17%) available in small quantities fit wearers whose size was in shortest supply. This scarcity led to the evaluation and purchase of a new respirator prototype for small N-95 wearers, which was an important success of our team’s work and for staff safety. Conclusions: Collaboration between teams from a variety of backgrounds, using both qualitative and quantitative data, resulted in a sustainable method for receiving, sorting, and evaluating donated N-95 respirators, ensuring the delivery of a steady supply of effective N-95 respirators to our staff. This quality-driven approach was an efficient and effective strategy to maintain our N-95 respirator supply during a pandemic driven global shortage.
Funding: No
Disclosures: None









